Scientist at work: To take atomic-scale pictures of tiny crystals, use a huge, kilometer-long synchrotron
- Written by Kerry Rippy, Ph.D. Candidate in Chemistry, Colorado State University
It’s 4 a.m., and I’ve been up for about 20 hours straight. A loud alarm is blaring, accompanied by red strobe lights flashing. A stern voice announces, “Searching station B. Exit immediately.” It feels like an emergency, but it’s not. In fact, the alarm has already gone off 60 or 70 times today. It is a warning, letting everyone in the vicinity know I’m about to blast a high-powered X-ray beam into a small room full of electronic equipment and plumes of vaporizing liquid nitrogen.
In the center of this room, which is called station B, I have placed a crystal no thicker than a human hair on the tip of a tiny glass fiber. I have prepared dozens of these crystals, and am attempting to analyze all of them.
 The author and her colleagues preparing crystalline samples to take to the synchrotron, in hopes of determining their atomic-level structures.
Courtesy of Kerry Rippy, CC BY-ND[1]
The author and her colleagues preparing crystalline samples to take to the synchrotron, in hopes of determining their atomic-level structures.
Courtesy of Kerry Rippy, CC BY-ND[1]
These crystals are made of organic semiconducting materials[2], which are used to make computer chips, LED lights, smartphone screens and solar panels. I want to find out precisely where each atom inside the crystals is located, how densely packed they are and how they interact with each other. This information will help me predict how well electricity will flow through them.
To see these atoms and determine their structure, I need the help of a synchrotron, which is a massive scientific instrument containing a kilometer-long loop of electrons zooming around at near the speed of light. I also need a microscope, a gyroscope, liquid nitrogen, a bit of luck, a gifted colleague and a tricycle.
Getting the crystal in place
The first step of this experiment involves placing the super-tiny crystals on the tip of the glass fiber. I use a needle to scrape a pile of them together onto a glass slide and put them under a microscope. The crystals are beautiful – colorful and faceted like little gemstones. I often find myself transfixed, staring with sleep-deprived eyes into the microscope, and refocusing my gaze before painstakingly coaxing one onto the tip of a glass fiber.
Once I’ve gotten the crystal attached to the fiber, I begin the often frustrating task of centering the crystal on the tip of a gyroscope inside station B. This device will spin the crystal around, slowly and continuously, allowing me to get X-ray images of it from all sides.
 On the left is the gyroscope, designed to rotate the crystal through a series of different angles as the X-ray beam hits it. Behind it is the detector panel which records the diffraction spots. On the right is a zoomed in picture of a single crystal, mounted on a glass fiber attached to the tip of the gyroscope.
Kerry Rippy, CC BY-ND[3]
On the left is the gyroscope, designed to rotate the crystal through a series of different angles as the X-ray beam hits it. Behind it is the detector panel which records the diffraction spots. On the right is a zoomed in picture of a single crystal, mounted on a glass fiber attached to the tip of the gyroscope.
Kerry Rippy, CC BY-ND[3]
As it spins, liquid nitrogen vapor is used to cool it down: Even at room temperature, atoms vibrate back and forth, making it hard to get clear images of them. Cooling the crystal to minus 196 degrees Celsius, the temperature of liquid nitrogen, makes the atoms stop moving so much.
X-ray photography
Once I have the crystal centered and cooled, I close off station B, and from a computer control hub outside of it, blast the sample with X-rays. The resulting image, called a diffraction pattern, is displayed as bright spots on an orange background.
 This is a diffraction pattern that results when you shoot an X-ray beam at a single crystal.
Kerry Rippy, CC BY-ND[4]
This is a diffraction pattern that results when you shoot an X-ray beam at a single crystal.
Kerry Rippy, CC BY-ND[4]
What I am doing is not very different from taking photographs with a camera and a flash. I’m about to send light rays at an object and record how the light bounces off it. But I can’t use visible light to photograph atoms – they’re too small, and the wavelengths of light in the visible part of the spectrum are too big. X-rays have shorter wavelengths, so they will diffract, or bounce off atoms.
However, unlike with a camera, diffracted X-rays can’t be focused with a simple lens. Instead of a photograph-like image, the data I collect are an unfocused pattern of where the X-rays went after they bounced off the atoms in my crystal. A full set of data about one crystal is made up of these images taken from every angle all around the crystal as the gyroscope spins it.
Advanced math
My colleague, Nicholas DeWeerd[5], sits nearby, analyzing data sets I’ve already collected. He has managed to ignore the blaring alarms and flashing lights for hours, staring at diffraction images on his screen to, in effect, turn the X-ray images from all sides of the crystal into a picture of the atoms inside the crystal itself.
In years past, this process might have taken years of careful calculations done by hand, but now he uses computer modeling to put all the pieces together. He is our research group’s unofficial expert at this part of the puzzle, and he loves it. “It’s like Christmas!” I hear him mutter, as he flips through twinkling images of diffraction patterns.
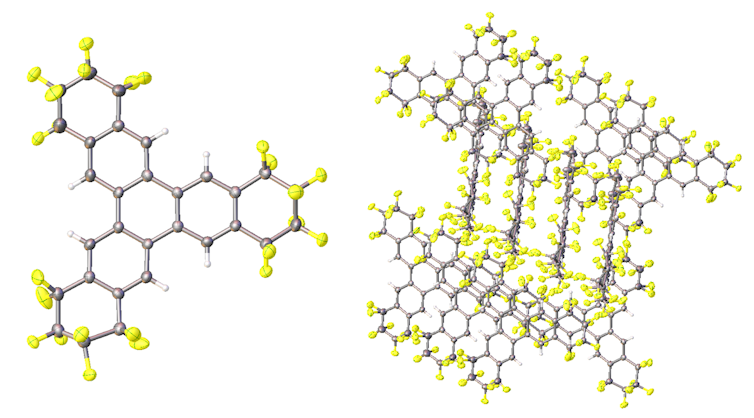 Solving a set of diffraction patterns produces an atomic-level picture of a crystal, showing individual molecules (left) and how they pack together to form a crystalline structure.
Kerry Rippy, CC BY-ND[6][7]
Solving a set of diffraction patterns produces an atomic-level picture of a crystal, showing individual molecules (left) and how they pack together to form a crystalline structure.
Kerry Rippy, CC BY-ND[6][7]
I smile at the enthusiasm he’s managed to maintain so late into the night, as I fire up the synchotron to get my pictures of the crystal perched in station B. I hold my breath as diffraction patterns from the first few angles pop up on the screen. Not all crystals diffract, even if I’ve set everything up perfectly. Often that’s because each crystal is made up of lots of even smaller crystals stuck together, or crystals containing too many impurities to form a repeating crystalline pattern that we can mathematically solve.
If this one doesn’t deliver clear images, I’ll have to start over and set up another. Luckily, in this case, the first few images that pop up show bright, clear diffraction spots. I smile and sit back to collect the rest of the data set. Now as the gyroscope whirls and the X-ray beam blasts the sample, I have a few minutes to relax.
I would drink some coffee to stay alert, but my hands are already shaking from caffeine overload. Instead, I call over to Nick: “I’m gonna take a lap.” I walk over to a group of tricycles sitting nearby. Normally used just to get around the large building containing the synchrotron, I find them equally helpful for a desperate attempt to wake up with some exercise.
As I ride, I think about the crystal mounted on the gyroscope. I’ve spent months synthesizing it, and soon I’ll have a picture of it. With the picture, I’ll gain understanding of whether the modifications that I have made to it, which make it slightly different than other materials I have made in the past, have improved it at all. If I see evidence of better packing or increased intermolecular interactions, that could mean the molecule is a good candidate for testing in electronic devices.
Exhausted, but happy because I’m collecting useful data, I slowly pedal around the loop, noting that the synchrotron is in high demand. When the beamline is running, it is used 24/7, which is why I’m working through the night. I was lucky to get a time slot at all. At other stations, other researchers like me are working late into the night.
Taking a tricycle for a ride at the Advanced Photon Source.References
- ^ CC BY-ND (creativecommons.org)
- ^ organic semiconducting materials (theconversation.com)
- ^ CC BY-ND (creativecommons.org)
- ^ CC BY-ND (creativecommons.org)
- ^ Nicholas DeWeerd (www.linkedin.com)
- ^ Kerry Rippy (doi.org)
- ^ CC BY-ND (creativecommons.org)
Authors: Kerry Rippy, Ph.D. Candidate in Chemistry, Colorado State University